Adipose Tissue and its dysfunctions in obesity
Your fat cells control your vital organs
Long relegated to the role of a mere fat reserve, adipose tissue is now recognized as a dynamic, complex, and endocrine organ that plays a central role in regulating energy metabolism and numerous physiological functions. The human body stores energy in the form of triglycerides within adipocytes (fat cells) to provide a mobilizable caloric reserve when needed. However, far from being inert, adipose tissue communicates with other organs via hormones and cytokines (known as adipokines), thereby modulating appetite, energy expenditure, insulin sensitivity, and even immune responses.
There are several types of adipose tissue with distinct properties, ranging from white fat that stores energy to brown fat that dissipates energy as heat. These fat deposits are distributed across different regions of the body, and this distribution strongly influences metabolic health: fat located around the viscera does not have the same effects as that located under the skin. Furthermore, adipose tissue exhibits remarkable plasticity. It can change in size, cellular structure, and function in response to the body’s conditions, such as during weight gain, exposure to cold, or pregnancy. This ability to adapt, or remodeling, is central to its healthy functioning but also to its dysfunction.
Indeed, in pathological conditions such as obesity (characterized by a chronic excess of calories stored as fat), adipose tissue can lose its adaptive capacity and become dysfunctional. “Diseased” adipose tissue results in overloaded and stressed adipocytes, a chronic inflammatory state, tissue fibrosis, and altered hormonal secretions. These dysfunctions directly contribute to complications of obesity such as insulin resistance, type 2 diabetes, non-alcoholic fatty liver disease, or cardiovascular disease. Conversely, maintaining healthy, functional adipose tissue appears to be a protective factor; indeed, it is possible to speak of metabolically healthy obesity in certain individuals whose adipose tissue, although abundant, manages to remodel itself without causing major metabolic disorders (at least for a certain period of time).
In the face of theglobal obesity epidemic, understanding how adipose tissue functions is a crucial scientific and medical challenge. This dossier offers a critical synthesis of current knowledge regarding the different types of adipose tissue and their distribution, adipocyte plasticity and its mechanisms, the endocrine functions of this organ, as well as the dysregulations of adipose tissue observed in obesity. We will also address the clinical implications of these concepts and emerging therapeutic approaches. The goal is to provide a clear and nuanced understanding of the role of adipose tissue in metabolic health, highlighting the main explanatory models, converging or contradictory research findings, and unresolved questions.
The Different Types of Adipose Tissue
Not all adipose tissues are the same: there are several types, defined by their cellular composition, location, and specific functions. Traditionally, we distinguish between white adipose tissue, brown adipose tissue, and an intermediate form known as beige adipose tissue. Each plays a specific role in the body.

Adipocyte blanc 
Adipocyte brun 
Adipocyte beige
White adipose tissue
White adipose tissue is the most abundant in adults. It is what is generally referred to when people talk about “body fat.” It consists mainly of white adipocytes, large cells (50 to 150 µm in diameter) containing a single large lipid droplet that occupies most of the cellular space. This so-called unilocular morphology is adapted to the primary function of white adipose tissue: storing triglycerides during periods of energy surplus, then releasing them in the form of fatty acids when the body needs them (fasting, prolonged exercise, exposure to cold). The white adipocyte is thus a cell specialized in energy storage and lipolysis (fat breakdown) on demand.
Beyond this role as a calorie reservoir, white adipose tissue is an important endocrine organ. It continuously secretes numerous signaling molecules called adipokines, which act locally or at a distance on the brain, liver, muscles, immune system, etc. Among the hormones produced by white fat are leptin (which informs the brain of energy reserves and tends to reduce appetite) and adiponectin (which increases insulin sensitivity and exerts anti-inflammatory and cardioprotective effects). White adipose tissue also produces pro-inflammatory cytokines (TNF-α, IL-6, etc.) , partly via resident immune cells, especially when adipocytes are hypertrophied. We will return to these endocrine functions in more detail later. Let us simply note here that white adipose tissue, far from being a passive tissue, communicates with the entire body and actively contributes to metabolic homeostasis.
In a healthy adult, white fat accounts for the majority of total fat mass and is located primarily under the skin (subcutaneous fat) and around the abdominal organs (visceral fat). It is also found in other specific locations (eye sockets, palms, soles of the feet, etc.) where it serves as a mechanical protective cushion. In summary, white adipose tissue is the body’s primary site for lipid storage and a major endocrine actor. It maintains energy balance by storing excess energy and redistributing it as needed, while sending hormonal signals regarding the state of reserves.
Brown adipose tissue
Brown adipose tissue is a very different type of fat, specialized in heat production rather than storage. It derives its name from its brownish color due to its high content of mitochondria (cellular organelles containing iron, hence the brown color). Brown adipocytes are smaller than white ones and contain multiple small lipid droplets (multilocular morphology). Most importantly, they express large amounts of a unique mitochondrial protein called UCP1 (uncoupling protein 1) or thermogenin, which bypasses normal ATP production in the mitochondria to directly generate heat. This process, called adaptive thermogenesis, allows for calorie burning by dissipating energy as heat, particularly in response to cold.
In humans, brown adipose tissue is abundant in newborns (particularly between the shoulder blades, around the neck, and near large blood vessels) to maintain body temperature. It was long believed that it disappeared almost entirely by adulthood. However, in 2009, several studies using positron emission tomography (PET scans) demonstrated the presence of active brown fat in some adults, located mainly in the cervical region, the shoulders, and along the spine. In a lean adult exposed to cold, these brown fat deposits capture glucose and burn fatty acids to produce heat, thereby contributing to energy expenditure. On the other hand, the amount of brown adipose tissue in adults varies greatly and tends to decrease with age and with excess weight (obese or elderly individuals have less of it, or it remains inactive).
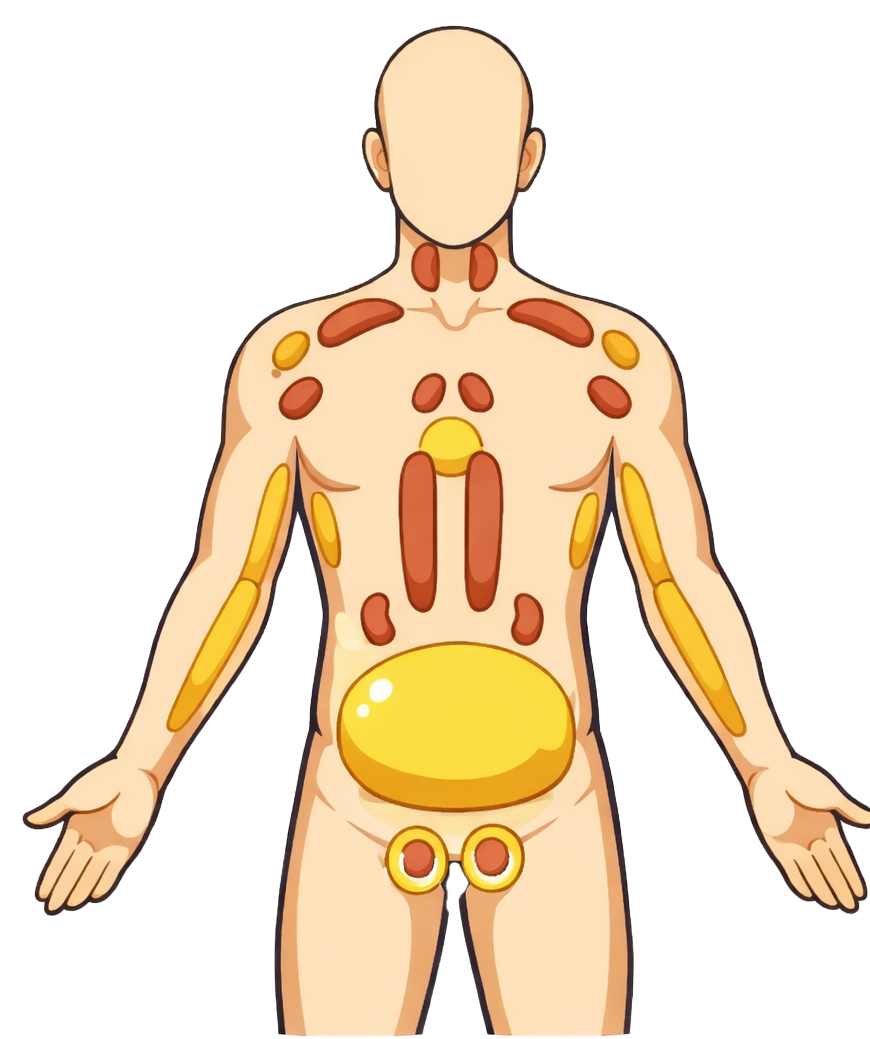
The primary physiological role of brown adipose tissue is to maintain body heat in cold conditions. It is activated under the control of the sympathetic nervous system (catecholamines). In addition to thermogenesis, BAT also has emerging endocrine functions: it secretes bioactive peptides and lipids (sometimes called “batokines”) that could influence overall metabolism, although these aspects are still under investigation. Overall, brown fat can be viewed as a biological “radiator,” converting energy into heat, in contrast to white fat, which acts more like an “energy reservoir of energy. This functional contrast makes it an attractive research target for combating obesity, by seeking to stimulate energy expenditure via brown tissue. However, in adult humans, the mass of this brown adipose tissue is low, and its overall metabolic impact remains limited compared to white fat.
Beige adipose tissue
Between white and brown, there exists a third category of adipocytes: beige adipocytes (sometimes called brite adipocytes, short for brown in white). This is not a distinct anatomical adipose tissue, but rather brown cells scattered within white adipose tissue. Indeed, under certain conditions, adipocytes that are halfway between white and brown appear in subcutaneous white fat deposits. These beige adipocytes exhibit intermediate characteristics: they possess multiple lipid droplets and mitochondria containing thermogenin (like brown fat), but coexist alongside classic unilocular white adipocytes within the same tissue.
The phenomenon of these induced brown adipocytes appearing within white fat is called browning (literally, the browning of white adipose tissue) or beiging. External stimuli such as prolonged exposure to cold or β -adrenergic stimulation (catecholamines) can trigger this browning: the white adipose tissue then develops a fraction of cells capable of thermogenesis, increasing energy expenditure. For example, studies show that a few weeks of moderate cold exposure in humans can increase the mass and activity of brown/beige adipose tissue, an improvement that regresses upon return to a warm environment. Similarly, certain hormonal factors linked to physical exercise or diet could induce browning. In this context, irisin, a myokine released by muscle during exercise, is notably the subject of extensive study as a possible trigger for browning, although the results are still debated.
Beige adipocytes express UCP1 and can, like brown adipocytes, burn fat to produce heat. However, they differ from classic brown adipocytes in terms of development and molecular composition. While brown fat in newborns derives from a precursor lineage shared with muscle cells, beige adipocytes appear rather from precursors present in white tissue, capable of differentiating into brown-type adipocytes upon stimulation. Some mature white adipocytes could even convert into beige adipocytes via a process of transdifferentiation, changing their phenotype. This process is reversible: if the stimulus disappears (for example, by returning to a warm temperature), beige adipocytes can either revert to a white phenotype (losing their excess mitochondria and UCP1), or disappear through apoptosis. This flexibility clearly illustrates the cellular plasticity of adipose tissue.
Beige adipose tissue is of great interest to researchers, as increasing the proportion of “good” brown/beige fat at the expense of white fat could theoretically help burn excess energy in overweight individuals. Studies in mice have shown that activating browning improves carbohydrate metabolism and insulin sensitivity. However, in humans, we do not yet know how to safely and effectively induce massive, sustained browning. The therapeutic potential of beige tissue therefore remains to be confirmed, but it is a fine example of adipose tissue’s ability to remodel its functional phenotype according to the body’s needs.
| Comparison of the main types of adipocytes in humans. | |||
|---|---|---|---|
| Characteristic | White adipocyte | Brown adipocyte | Beige adipocyte |
| Cell morphology | Large unilocular cell (1 large lipid droplet) | Multilocular cell (multiple small droplets) | Multilocular (within white tissue) |
| Typical size | 50–150 µm (varies depending on nutritional status) | 20–50 µm (smaller than white) | Intermediate (size similar to brown) |
| Number of mitochondria | Low (few mitochondria) | Very abundant (rich in mitochondria containing UCP1) | Abundant (expresses UCP1 upon stimulation) |
| Main function | Energy storage (triglycerides) | Energy expenditure via thermogenesis (heat production) | Induced thermogenesis (depends on stimulation, e.g., cold) |
| Location | Throughout the body (subcutaneous, visceral, etc.) | Specific areas (infants: interscapular; adults: neck, mediastinum, etc.) | Integrated into white tissue (mainly subcutaneous) |
| Endocrine role | Yes – production of leptin, adiponectin, etc. (numerous adipokines) | Yes – production of factors (e.g., FGF21) but endocrine role less well understood | Yes – similar to brown fat when activated (secretion of certain adipokines) |
| Activation | Energy stimuli (calorie excess → storage, deficit → lipolysis) | Cold, β-adrenergic stimulation (sympathetic) | Cold, β-adrenergic stimulation, hormones and exercise |
| Metabolic impact | Essential calorie reserve but excess → metabolic syndrome | Increases energy expenditure (may protect against obesity) | Slightly increases energy expenditure (if activated) |
In summary, white fat is the primary adipose compartment in adults, dedicated to energy storage and hormonal signaling. Brown fat, more abundant in infants, allows energy to be expended to produce heat. As for beige adipocytes, they represent an adaptive potential of white adipose tissue, giving it the ability to burn calories under certain conditions. The coexistence of these cell types underscores that adipose tissue is a multifunctional and adaptable organ, far removed from the monolithic view held in the past.
Anatomical Distribution and Functional Diversity of Adipose Deposits
Beyond differences in cell types, the anatomical location of adipose tissues plays a decisive role in their functions and their impact on health. The human body has numerous distinct adipose deposits, distributed from head to toe, which differ in their anatomy, vascularization, innervation, and interactions with neighboring organs. This section explores the anatomical diversity of adipose tissue and the functional consequences of fat distribution.
Subcutaneous fat vs. visceral fat
The most common distinction in medicine and sports science contrasts subcutaneous fat subcutaneous fat (located under the skin) and visceral fat (accumulated within the abdominal cavity, around the organs). Both are white adipose tissue, but their behavior and metabolic effects differ significantly.
It is important to note that the subcutaneous/visceral distinction does not cover all nuances. For example, deep abdominal subcutaneous fat (located beneath the muscle layer) may exhibit intermediate metabolic behavior. Nevertheless, this distinction largely explains why two people with the same BMI may have very different disease risks: it all depends on whether excess fat is stored as “benign” subcutaneous fat or “toxic” visceral fat.
In clinical and sports practice, this distribution is assessed by measuring waist circumference or the waist-to-hip ratio. A high waist circumference indicates visceral fat accumulation and an increased cardiometabolic risk, even in individuals who do not appear obese (this is the case with the TOFI profile, i.e., Thin Outside, Fat Inside). Conversely, an overweight individual with a more gynoid distribution (wide hips, moderate waist) may have a lower risk of diabetes. Thus, the anatomical distribution of adipose tissue is a key factor in its impact on health.
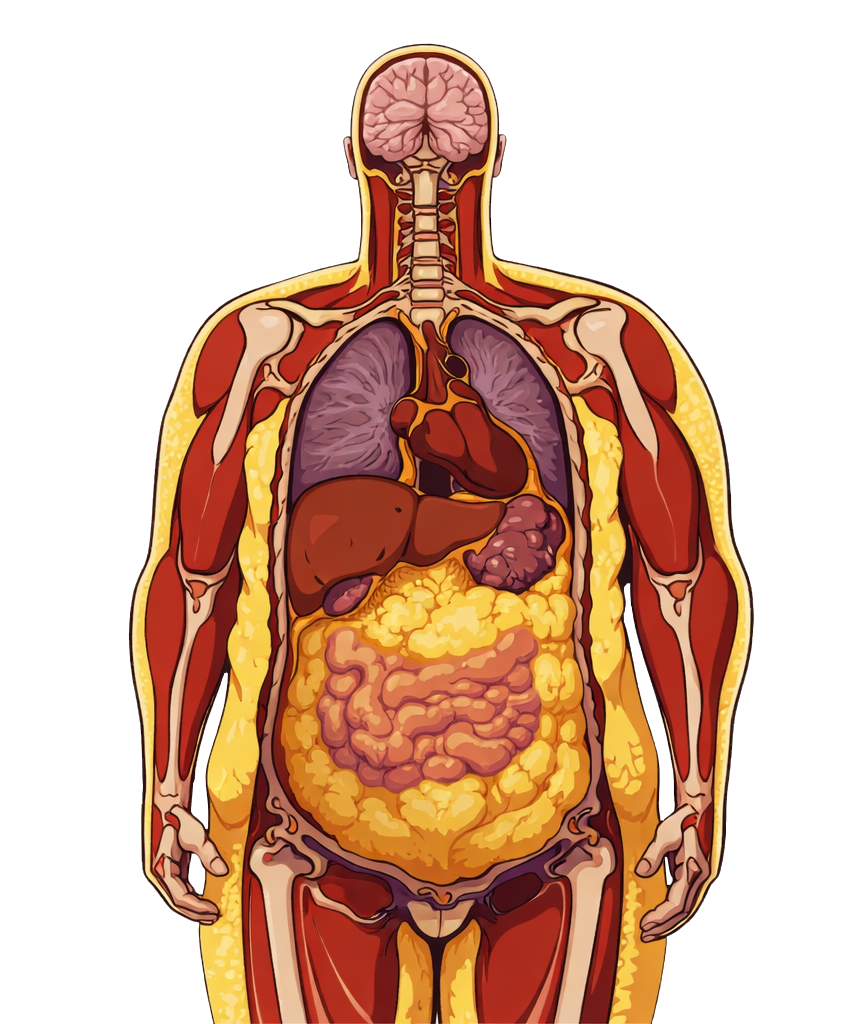
MUO (Metabolically Unhealthy Obesity) 
TOFI (Thin Outside, Fat Inside)
Why is visceral fat more harmful than subcutaneous fat?
Several non-mutually exclusive hypotheses exist:
Specific fat deposits and unique roles
In addition to subcutaneous and visceral fat, the body contains more subtle fat deposits that often perform essential, sometimes localized and non-traditional functions. Here are a few notable examples:
This list is not exhaustive: we should also mention peritoneal fat (which lines the inside of the abdominal cavity) , perirenal fat (around the kidneys, serving a supportive and protective role), and orbital fat (behind the eyeballs, supporting eye movements) . Each of these deposits may have specific mechanical and metabolic functions.
The anatomical diversity of adipose tissue explains why we can speak of “adipose tissues” in the plural. Far from being homogeneous, the human body’s adipose system is a true mosaic, with each local deposit able to adapt to the needs of its immediate environment while contributing to overall balance. Some deposits perform vital functions (thermogenesis, local energy support, immune regulation of the skin, etc.), while others serve as reserves that can be mobilized when necessary. But this compartmentalization also has a downside: depending on where excess fat is stored, the consequences will not be the same. For example, 1 kg of excess fat located around the liver or heart will have far more harmful consequences than 1 kg distributed under the skin of the thighs. This concept is essential for assessing an individual’s metabolic risk and for tailoring weight management strategies. It also explains certain apparent contradictions in studies: you can have a “normal” BMI but hidden excess visceral fat and be at risk for diabetes, or conversely, be obese but with good fat distribution and functional adipose tissue, and remain in better metabolic health (at least temporarily).
In short, location matters just as much as quantity. Understanding the specific characteristics of each fat deposit helps interpret the body’s adaptations (to exercise, cold, overeating) and the pathological mechanisms of obesity. Let’s now look at how these different deposits can adapt (plasticity) or, conversely, malfunction depending on the stresses placed on them.
Plasticity and Remodeling of Adipose Tissue
Adipose tissue is often described as highly plastic, meaning it is capable of altering its structure and cellular composition in response to changes in the body or the environment. This plasticity manifests in multiple ways: expansion or reduction of fat mass, cellular remodeling (hyperplasia, hypertrophy, differentiation), changes in vascular architecture and the extracellular matrix, alterations in the tissue’s immune profile, etc. It allows adipose tissue to fulfill its functions under a wide variety of conditions (starvation or abundance, cold or heat, growth, pregnancy, etc.), but it also has limits; exceeding these limits leads to dysfunction.
Browning / Beiging: The Emergence of Beige Adipocytes
One of the most striking examples of adipose tissue plasticity is the phenomenon of browning of white adipose tissue, already introduced above. As a reminder, this refers to the ability of white tissue to acquire characteristics of brown tissue under the influence of certain stimuli, through the emergence of thermogenic beige adipocytes. This process illustrates the phenotypic plasticity of adipocytes: a storage cell can, within certain limits, reprogram itself into a fat-burning cell.
Browning is primarily induced by the activation of the sympathetic nervous system, typically during prolonged exposure to cold. The sympathetic nerves innervating adipose tissue release norepinephrine, which binds to the β-3 adrenergic receptors on white adipocytes. This triggers a signaling cascade (cAMP-PKA pathway) leading to the expression of genes characteristic of brown adipocytes (such as UCP1) in certain cells of the white fat deposit. These cells increase their number of mitochondria, fragment their large lipid droplets into smaller droplets, and begin to oxidize fatty acids to produce heat instead of storing them. At the same time, mesenchymal precursors present in adipose tissue can be recruited to differentiate directly into brown-type adipocytes. The macroscopic result, after a few weeks of cold exposure, is that the previously homogeneous white adipose tissue takes on a browner color and exhibits increased metabolic activity: this is beige adipose tissue.
Browning is not, however, a permanent state: it depends on continuous stimulation. If the stimulus is removed (for example, by warming the body), beige adipocytes gradually lose their expression of UCP1 and revert to being “white” from a functional standpoint. This rapid reversibility has been demonstrated in animals and in studies of humans exposed to cold: adipose tissue biology adapts within a few days or weeks in either direction. This suggests that beige adipocytes derived from white ones do not necessarily die when the cold ceases, but can redifferentiate toward a storage phenotype. This ability is an active area of research, as understanding how to maintain browning could be useful for increase energy expenditure.
Apart from cold, other factors can modulate browning:
The concept of browning has raised high hopes in the fight against obesity, as it offers the prospect of dissipating excess energy by redirecting the function of adipose tissue. However, it must be kept in mind that even when activated, brown/beige tissue in adults accounts for only a small fraction of total body fat. The additional caloric expenditure achievable through browning (a few dozen kcal per day in realistic studies) is insufficient on its own to offset a high-calorie diet or induce significant weight loss. Furthermore, increasing thermogenesis is not without risk (cardiovascular stress, feelings of cold, etc.) . Thus, browning is an interesting metabolic tool, especially for improving blood glucose and blood lipid levels (by burning excess glucose and fatty acids), but it is not a miracle cure for obesity. Its clinical value may lie in complementing other approaches, for example to help obese patients manage their diabetes by increasing their energy expenditure without intense physical exertion.
In summary, browning illustrates the functional plasticity of adipose tissue: within a few days, the same area of fat can change its function (shifting from storage to dissipative energy expenditure) without changing location. This occurs through the reprogramming of certain adipocytes and the recruitment of new brown adipocytes. This process, beneficial under certain conditions (cold, exercise), demonstrates the extraordinary adaptability of adipose tissue to the body’s energy needs.
Hyperplasia vs hypertrophy: modes of adipose tissue expansion
When an individual consumes more calories than they expend over an extended period, the excess energy must be stored somewhere. White adipose tissue is the primary site of this storage. Fat mass gain involves two distinct cellular mechanisms: hypertrophy and adipocyte hyperplasia. The balance between these two modes of expansion largely determines the health of adipose tissue and, consequently, overall metabolic health.

Hyperplasia 
Hypertrophy
These two mechanisms can, of course, coexist, but their relative proportions differ depending on the individual, the fat deposits, and the circumstances. This proportion has significant consequences.
Excessive hypertrophy of adipocytes is associated with adipose tissue dysfunction. Hypertrophy is accompanied by an alteration in the adipocyte secretion profile. Adipocyte size is a crucial factor: hypertrophied adipocytes have impaired metabolism and secretion, promoting insulin resistance* “. In short, the larger the adipocytes, the more they malfunction**.
Conversely, adipocyte hyperplasia is often considered a “healthy” expansion of adipose tissue. By increasing the number of cells, the tissue can store more lipids without each individual adipocyte reaching a critical size. It is better to have 100 adipocytes filled to 50% of their capacity than 50 adipocytes filled to 100% . Smaller, more numerous adipocytes maintain better insulin sensitivity and a more favorable hormonal secretion profile (more adiponectin, fewer inflammatory cytokines). Studies show that obese individuals who maintain moderately sized adipocytes, thanks to active hyperplasia, have a healthier metabolic profile than those whose adipocytes reach extremely large volumes .
This relates to the concept of the “expansion capacity” of adipose tissue: when capacity is good (many new adipocytes can be formed) , excess energy is safely stored in adipose tissue, protecting other organs. But if expansion capacity is limited (not enough new cells), then existing adipocytes swell beyond reasonable limits, and the excess “spills over” into other organs (liver, muscles). This is where complications begin.
One might then think that promoting adipocyte hyperplasia is always beneficial. It’s not that simple. First, not everyone has the same reserve of preadipocytes or the same genetic predisposition to hyperplasia. In fact, genetic factors influence the development of adipose tissue. For example, some people have more adipocytes from birth (babies with more fat cells will have less need for hypertrophy later on). Furthermore, once the number of adipocytes has increased (during childhood or adolescence, periods conducive to hyperplasia), it tends to remain high even in the event of weight loss. Isotopic studies have shown that the number of adipocytes in adults is relatively fixed; we renew about 10% of our adipocytes each year, but the total number changes little, except in cases of massive weight gain. This means that adipocyte hyperplasia, once it has occurred, leaves a legacy: if an individual has increased their number of adipocytes during a period of obesity, they will retain these adipocytes even after losing weight (though they will be empty). However, a high number of empty adipocytes can facilitate rapid weight regain, as these “underfilled” cells potentially send signals indicating a need to be filled (low leptin per adipocyte, etc.). This phenomenon is often cited to explain the frequent weight rebound following a restrictive diet. It is, in a way, the negative counterpart of hyperplasia.
Despite this, from a purely metabolic standpoint, at equal weight, it is better to have more small cells. Arner and colleagues (2010), for example, showed that obese individuals with hypertrophied adipocytes exhibited much greater insulin resistance than obese individuals with the same BMI who had smaller and more numerous adipocytes. Similarly, certain antidiabetic treatments such as thiazolidinediones (TZDs) exploit hyperplasia: these PPAR-γ agonists increase the differentiation of new adipocytes, which distributes lipid storage more effectively and improves insulin sensitivity despite potential additional weight gain.
It should be noted that the capacity for hyperplasia varies depending on the fat deposits. Subcutaneous fat has a good capacity to generate new adipocytes, especially in the lower body. In contrast, visceral fat has fewer available or activatable preadipocytes, which would explain why visceral expansion is very quickly accompanied by hypertrophy and dysfunction. This observation is consistent with the fact that a chronic caloric excess first results in an expansion of the subcutaneous compartment; then, when this reaches saturation, fat overflows into the visceral space and organs (this is the theory of lipid overflow). Thus, the inability of subcutaneous tissue to recruit enough adipocytes to store excess fat would be a trigger for pathological visceral and ectopic fat storage.
In summary, adipocyte hyperplasia is a sign of positive plasticity, allowing for expandable and metabolically neutral storage, whereas excessive hypertrophy indicates that the adaptive capacity of adipose tissue has been exceeded. Promoting hyperplasia (within certain limits) could be an approach to maintaining good metabolic health despite moderate weight gain; this is what is observed in metabolically healthy obese individuals. Conversely, when hypertrophy dominates without compensatory hyperplasia, adipose tissue becomes the driving force behind metabolic disorders. The inflammatory, endocrine, and metabolic consequences of this shift will be discussed in the section on adipocyte dysfunction.
Plasticity in Various Physiological and Pathological Contexts
To complete this overview of adipose plasticity, it is useful to illustrate how adipose tissue behaves in various everyday situations or pathological conditions. We have already discussed the case of expansion in obesity at length. Let us briefly address other contexts:
Prolonged fasting and weight loss
During severe caloric restriction, adipose tissue loses volume through triglyceride mobilization (increased lipolysis). Adipocytes shrink (sometimes to sizes smaller than normal). However, the number of adipocytes does not drop drastically: the empty cells persist but enter a “rest.” In individuals who have undergone massive weight loss (strict dieting or bariatric surgery), adipose tissue consists of numerous small, sometimes atrophic adipocytes, with residual fibrosis. The inflammatory profile improves significantly (fewer M1 macrophages), but the fibrosis accumulated during obesity disappears only partially. The tissue therefore retains structural sequelae, which could limit its subsequent re-expansion or, conversely, facilitate adipose regeneration (a matter of debate). Fasting also alters cellular composition: for example, an increase in brown/beige adipocytes is observed (in animals) despite weight loss, perhaps to compensate for the decrease in thermogenesis due to the loss of white adipose tissue.
Exercise and Training
Regular physical exercise induces systemic adaptations, including in adipose tissue. On the one hand, exercise reduces fat mass by increasing energy expenditure and lipid oxidation. It primarily targets visceral and ectopic fat deposits, which generally decrease the fastest. On the other hand, exercise improves the vascularization of adipose tissue (via enhanced perfusion) and shifts the profile of immune cells toward an anti-inflammatory state. Studies have shown that after several weeks of training, adipocyte secretion is altered: more adiponectin, less TNF-α, for example. It has also been suggested that exercise may moderately stimulate browning, particularly through irisin, although the effect is mild in humans. Thus, exercise confers a healthier and more plastic profile on adipose tissue: adipocytes become smaller and more insulin-sensitive, the matrix is less rigid, and inflammation decreases. This contributes to improved insulin sensitivity and reduced metabolic risk in active individuals. Another important aspect is that exercise increases the ability of adipocytes to rapidly recycle lipids between storage and utilization. In endurance athletes, adipocytes are trained to release fatty acids during exercise and then refill without dysfunction, demonstrating the tissue’s high metabolic flexibility.
Pregnancy and the Postpartum Period
Pregnancy is a hyperanabolic state in which the body stores energy for the fetus and future breastfeeding. In pregnant women, there is a programmed increase in fat mass, with marked adipocyte hyperplasia in certain regions (buttocks, thighs, arms). This is a physiological phenomenon regulated by hormones. Adipose tissue thus prepares to mobilize reserves during late pregnancy and breastfeeding. Subsequently, during breastfeeding, prolactin and other hormones induce fat mobilization (increased lipolysis) to supply fatty acids to the milk, which often leads to gradual weight loss in the mother. The remodeling of adipose tissue during and after pregnancy is fascinating: not only do adipocytes change in size, but as seen earlier, in the mammary gland, they can transdifferentiate into milk-secreting cells. This is an extreme example of plasticity, where the adipocyte temporarily relinquishes its storage function to transform into another functional cell. After weaning, most of these cells will revert to adipocytes. This cyclical nature can leave lasting effects (for example, the number of mammary adipocytes may increase after each lactation cycle). From a general metabolic perspective, pregnancy induces a certain degree of physiological insulin resistance (to conserve glucose for the fetus), and adipose tissue contributes to this by altering its secretions: increased leptin, resistin, etc. This is normal and reversible after childbirth for most women, proof that these adipocyte alterations were indeed programmed and not pathological (unless gestational diabetes occurs, possibly indicating that the tissue’s adaptive capacity has been exceeded).
Aging
With age, fat distribution tends to become more centralized (more visceral fat, less peripheral subcutaneous fat, particularly in postmenopausal women). Aging adipose tissue loses plasticity: it becomes less hyperplastic, more fibrous, and recovers less effectively after stress. There is a decrease in the fraction of preadipocytes capable of differentiating. Furthermore, brown adipose tissue declines with age, which reduces adaptive thermogenesis (leading to a tendency to gain weight more easily). The immune microenvironment of adipose tissue also exhibits increasing low-grade chronic inflammation (a phenomenon known as “inflammaging”). All these changes contribute to a higher incidence of insulin resistance in older adults with the same BMI. In other words, an older person’s adipose tissue is less tolerant of caloric excess:
it becomes dysregulated more quickly. This underscores the importance of maintaining good physical fitness, which, as we have seen, helps preserve adipocyte plasticity through exercise and an appropriate diet.
Chronic Stress and Cortisol
Cortisol, the stress hormone, has a specific effect on fat distribution: it promotes the accumulation of visceral fat at the expense of peripheral subcutaneous fat (this is striking in Cushing’s syndrome, where trunk obesity is very pronounced, for example). Cortisol also partially inhibits the formation of new adipocytes (instead promoting the swelling of existing adipocytes). Chronic stress with elevated cortisol could therefore reduce hyperplastic plasticity and lead to visceral hypertrophy.
Cachectic conditions (cancer, AIDS)
In contrast to obesity, certain diseases cause severe depletion of adipose tissue, as in the case of cachexia (which is also accompanied by significant loss of muscle mass). In the case of cancer, tumor factors disrupt metabolism: they cause massive, insulin-resistant lipolysis , leading to rapid fat loss. Cachectic adipose tissue is characterized by inflammatory infiltration, adipocyte apoptosis, and a near absence of beige/brown adipocytes (paradoxically, the body shuts down adaptive thermogenesis to conserve energy, but basal energy expenditure remains high due to the tumor). This is an area where researchers are trying to understand how adipocyte plasticity (or its hijacking by the tumor) contributes to extreme weight loss.
Ultimately, these numerous examples demonstrate that adipose tissue is far from static. It reacts sensitively to nutritional, hormonal, and environmental states. This adaptive plasticity is generally beneficial, allowing the body to store and mobilize energy appropriately in every situation. However, there are breaking points: when demands exceed the tissue’s capacity to tissue to adapt, plasticity diminishes and the tissue shifts into a harmful state (chronic inflammation, fibrosis, cell death). The balance between successful remodeling and failed remodeling largely determines whether weight gain will remain benign or result in a metabolic disorder.
Dysfunctions of adipose tissue in obesity
Obesity is not simply an excess of body fat; it is also a state in which fat behaves differently, abnormally from a biological standpoint. It is sometimes referred to as adipose tissue disease to describe the fact that, in obese individuals (especially those with abdominal obesity), adipose tissue exhibits characteristics of dysfunction. This manifests as:
These abnormalities within adipose tissue have serious systemic consequences: they play a central role in the development of type 2 diabetes, cardiovascular disease, hypertension, NAFLD, and other obesity-related complications. In this section, we will analyze the various aspects of dysfunctional adipose tissue in obese individuals and the mechanisms by which they contribute to disease.
From Adaptive Expansion to Pathological Expansion
Let’s break down the key events that mark the shift toward pathological expansion of adipose tissue. We can consider that there is a sort of tipping point where saturated adipose tissue ceases to be an ally of metabolism and becomes a disruptor. Several intertwined phenomena contribute to this tipping point: exceeding safe storage capacity, hypoxia, vascular insufficiency, mechanical stress, alterations in the extracellular matrix, and fibrosis.
Safe storage capacity exceeded: This concept means that healthy adipose tissue can store a certain amount of dietary fat surplus without harm; beyond a certain threshold, each additional lipid calorie to be stored would become “ dangerous” because it would contribute to adipose tissue dysfunction or be diverted to other organs. This threshold corresponds to the well-known adipocyte expansion limit mentioned earlier. When this limit is reached, complications begin to arise.
But what actually happens when adipose tissue is full? One of the first problems is insufficient vascularization. During rapid expansion, the growth of adipocytes can exceed the tissue’s capacity to form new blood vessels to oxygenate them properly. Consequently, areas of chronic hypoxia are found in hypertrophic adipose tissue. Adipocytes in hypoxia trigger a stress response via the transcription factor HIF-1α (Hypoxia-Inducible Factor 1). However, in obese adipose tissue, it has been shown that HIF-1α does not merely induce compensatory angiogenesis: it also activates an entire pro-inflammatory and pro-fibrotic program. In particular, HIF-1 stimulates the expression of collagen and inhibitors of enzymes that degrade collagen, promoting the accumulation of extracellular matrix. The result is that hypoxia paradoxically leads to tissue fibrosis rather than an effective correction of the vascular deficit. This fibrosis creates rigid scar tissue that further limits the expansion of adipocytes (it is like a fibrous corset that constricts growing cells).
It is a vicious cycle: hypoxia leads to fibrosis, which causes a reduction in plasticity, resulting in even greater local hypoxia.
Some researchers therefore suggest that adipocyte hypertrophy is not problematic in itself; rather, it may be the fact that the adipocyte cannot hypertrophy further. It has been observed in mouse models that specifically knocking out HIF-1α in adipocytes reduces fibrosis in obese adipose tissue and improves insulin sensitivity, highlighting the importance of this pathogenic pathway.
At the same time, insufficient vascularization causes a nutrient supply problem: adipocytes at the periphery of the adipose lobule receive a priority supply of insulin and glucose, but those in the center lack them. This may contribute to the heterogeneity in size and function of adipocytes within the same deposit. Some remain insulin-sensitive (periphery), while others become insulin-resistant and lipolytic (hypoxic center) . This dysfunctional mosaic sustains abnormal fatty acid fluxes.
Mechanical stress on the matrix: As adipocytes grow larger, they exert mechanical pressure on neighboring structures and the extracellular matrix. The matrix (a network of collagen and proteoglycans) must stretch. If the adipocyte grows faster than the matrix can yield, there is an increase in tissue stiffness and negative feedback on the cells. Adipocytes and preadipocytes possess mechanical sensors (integrins, focal adhesions) that, when they detect a rigid substrate, send an anti-differentiation signal. Consequently, a stroma that is too fibrotic and rigid can block adipogenesis (because the cells sense that there is no longer room or flexibility to accommodate new cells). This locks the situation into a hypertrophic state. Furthermore, direct mechanical stress on an adipocyte (for example, two hypertrophic adipocytes compressing each other) can trigger the inflammasome signaling pathway (via stretch-sensitive ion channels), leading to the secretion of IL-1β, a pro-inflammatory cytokine. Thus, physically overloaded adipose tissue is tissue that sends out chemical distress signals.
The combination of hypoxia and mechanical stress leads to cell death: As mentioned, these combined factors often result in the death of some adipocytes. Hypoxia causes necrosis of the nucleus of an adipose lobule due to a local lack of ATP, and mechanical stress can rupture the cell membrane. Cell death (primarily necrotic) is a major trigger for inflammation: cellular contents are released (fatty acids, DNA, etc.) and act as danger signals for surrounding macrophages. We then move from simple moderate macrophage infiltration to full-blown inflammation (see next section). From a histological perspective, it is the massive appearance of macrophage crowns surrounding dead adipocytes that signals this shift. It is often at this stage that adipose tissue is considered to have entered a pathological state that fuels metabolic problems rather than preventing them.
In summary, as long as adipose tissue can remodel its infrastructure (vessels, matrix) and recruit new cells, it absorbs excess energy without much difficulty. When it can no longer do so, expansion occurs through extreme hypertrophy, causing hypoxia, fibrosis, cellular stress, and adipocyte death. These local abnormalities (microhypoxia, segmental fibrosis) are characteristic of obese and pathological adipose tissue and are rarely present in healthy adipose tissue, even when voluminous (for example, in a person gaining weight moderately, their subcutaneous fat enlarges but remains well-oxygenated and minimally fibrotic). Furthermore, the failure of adipose tissue to store fat leads to ectopic storage: when fat can no longer enter saturated adipocytes, excess lipids infiltrate other organs (liver, skeletal muscle, heart, pancreas…) where they will cause local dysfunction (hence the term lipotoxicity). Finally, note that visceral fat generally has a lower capacity for hyperplasia than subcutaneous fat, which is why it becomes dysfunctional more quickly in the event of excess. Also, the threshold at which problems arise differs from person to person.
Inflammation, Immunity, and Senescence: The Shift Toward Diseased Tissue
A defining feature of dysfunctional adipose tissue is the presence of chronic low-grade inflammation within it. This local adipocyte inflammation, once unrecognized, is now acknowledged as a central link connecting obesity to metabolic complications. In this section, we examine how the adipose tissue immune system shifts during pathological expansion, with the influx of pro-inflammatory macrophages, the involvement of other immune cells, the emergence of senescent cells, and how all of this fuels the vicious cycle of dysfunction.
Macrophages and Adipocyte Inflammation:
In healthy adipose tissue (e.g., in a lean individual), macrophages account for approximately 5–10% of adipose tissue cells, and most have an anti-inflammatory phenotype (known as M2 macrophages, involved in tissue homeostasis and repair). They clear debris and help maintain a favorable environment. In contrast, in obese and hypertrophied adipose tissue, the proportion of macrophages can rise to 50% of the cells in the vascular stroma! The majority of these macrophages then have a pro-inflammatory phenotype (so-called M1-type): they express markers such as CD11c and secrete TNF-α, IL-6, IL-1β, MCP-1, etc. Where do they come from? Adipocyte hypertrophy leads to increased release of MCP-1 (CCL2), a chemokine that recruits monocytes from the blood to adipose tissue. Once infiltrated, these monocytes differentiate into M1 macrophages. Furthermore, adipocyte death provides a powerful signal for resident macrophages to proliferate locally (via factors such as GM-CSF). This leads to the formation of “crowns” form around the dead adipocytes: lipid-laden macrophages that digest them. This process of phagocytosis of lipid remnants can transform macrophages into foam cells similar to those seen in atherosclerotic plaques, prompting them to produce more inflammatory cytokines and enzymes (collagenases, etc.).
M1 macrophages in adipose tissue thus become the primary source of local inflammatory cytokines, which also diffuse into the circulation. For example, IL-6 produced by adipose tissue contributes to elevated levels of C-reactive protein (CRP) -reactive protein), a marker of systemic inflammation that is elevated in obesity. TNF-α, for its part, interferes with insulin signaling (it phosphorylates IRS-1 at inhibitory serine residues), which reinforces insulin resistance in adipocytes (and in muscle via the circulation).
Other immune cells
Macrophages are not alone. As early as the initial stages of weight gain, T lymphocytes appear in adipose tissue to modulate the immune response. Studies in mice show that even even before the massive influx of macrophages, CD8+ T lymphocytes infiltrate the fat in response to these changes, and that by eliminating them, the arrival of macrophages can be delayed. These pro-inflammatory CD8+ T lymphocytes also produce IFN-γ and other signals that activate macrophages. CD4+ T cells (helper cells) can switch from an anti-inflammatory Treg profile to a pro-inflammatory Th1 profile in obesity. An increase in NK (Natural Killer) cells is also observed in obese fat tissue; these cells can kill stressed adipocytes (via NKG2D ligands) or stimulate macrophages. Mast cells (immune cells associated with ) also infiltrate the fat and release mediators that exacerbate inflammation. Even neutrophils make a transient appearance at the start of a high-calorie diet, releasing enzymes and free radicals. Thus, the entire innate and adaptive immune system converges on the pathologically expanding fat, as if it were damaged tissue requiring a defense/repair response.
Unfortunately, this chronic immune response becomes maladaptive. Instead of simply repairing the tissue, it causes further damage. Chronic inflammation promotes fibrosis (via factors such as TGF-β released by macrophages or mast cells, which stimulate fibroblasts to produce collagen). It generates oxidative damage: activated macrophages produce NO and superoxide, which can combine to form peroxynitrite, damaging neighboring cells. This oxidative stress can further impair the function of remaining adipocytes (for example, by oxidizing fatty acids, which can disrupt insulin signaling).
Cellular senescence
A phenomenon linking inflammation and loss of plasticity is the senescence of adipose tissue cells. Cellular senescence is a state in which a cell stops dividing and secretes a range of pro-inflammatory and pro-degradative factors (including pro-inflammatory cytokines known as SASP). In aging or obese adipose tissue, senescent adipocytes are found, as well as pre- senescent preadipocytes. These senescent cells emit signals (IL-6, IL-1β, MMPs, etc.) that amplify immune recruitment and tissue degradation. A senescent preadipocyte can no longer differentiate into an adipocyte, which reduces hyperplastic capacity. Furthermore, it can hinder its neighbors by releasing antagonistic factors (e.g., activin A, which inhibits adipogenesis). Thus, the accumulation of senescent cells in obese fat is an aggravating factor in dysfunction, locking the tissue into an inflammatory and rigid state. Studies in mice have shown that the pharmacological elimination of senescent cells ( via senolytics) in adipose tissue improved blood glucose levels and adipocyte function, highlighting their causal role.
We can now describe the complete vicious cycle that sets in within dysfunctional adipose tissue:
Adipocyte hypertrophy causes hypoxia and cell death, which attracts and activates pro-inflammatory macrophages that release TNF-α, IL-1β, etc., and which exacerbate insulin resistance in neighboring adipocytes and inhibit adipogenesis . The tissue becomes increasingly unable to store fat and grows in a disorderly manner, leading to the death of more adipocytes. Cytokines perpetuate fibrosis, yet chronic fibrosis and inflammation prevent any reversal of the process and further damage storage capacity, consequently leading to more circulating fat, and so on.
This adipocyte vicious cycle simultaneously fuels a systemic vicious cycle: adipocyte inflammation leads to moderate systemic inflammation (increased CRP, circulating cytokines) which causes hepatic and muscular insulin resistance, leading to hyperglycemia and hyperinsulinemia, which further stimulate hepatic lipogenesis and macrophage differentiation… We can see how everything is self-perpetuating, painting the picture of full-blown metabolic syndrome.
It is important to note that this inflammation is generally clinically silent (no pain, no fever), but it is biologically measurable (through markers such as CRP or plasma IL-6). Nor is it as intense as acute immune-mediated inflammation, but this constant low-grade inflammation is sufficient to disrupt target organs over time.
The direct consequences for adipose tissue itself are a loss of normal endocrine function. Reducing this inflammation is a viable therapeutic goal. Pilot studies have tested anti-inflammatory drugs (such as salicylate, a mild anti-inflammatory, or IL-1 antagonists) in obese diabetic patients, showing a modest improvement in blood glucose levels, confirming that targeting adipocyte inflammation can be helpful. However, completely eliminating inflammation is not straightforward, as it is an integrated response of the body to excess nutrients. The ideal approach is to act upstream by preventing adipocyte size from triggering this process, through early dietary and physical activity interventions.
In conclusion, the transition of adipose tissue into diseased tissue is characterized by the onset of chronic inflammation orchestrated by macrophages and other immune cells, as well as by the emergence of senescent adipocytes and progenitor cells. This inflammation triggers a cascade of local disturbances (fibrosis, ongoing cell death) that prevent the tissue from fulfilling its role, and simultaneously generate system-wide signals that impair insulin sensitivity and cardiovascular health. With this understanding, we can address the final aspect of adipocyte dysfunction: its role as an impaired endocrine organ, that is, how pathological adipose tissue negatively influences the rest of the body through its secretions and its inability to properly regulate overall metabolism.
Dysfunctional endocrine organ: the role of adipokines
The term adipokine refers to any bioactive substance released by adipose tissue that has a local or systemic effect. Since the discovery of leptin in 1994, dozens of adipokines have been identified, reflecting the versatility of adipose tissue as an endocrine gland.
In addition to being a site of energy storage, adipose tissue acts as a true endocrine organ. As such, it secretes a wide variety of hormones, cytokines, and mediators that convey messages to other organs. In a healthy state, it plays the role of a sensible metabolic conductor: it sends coherent hormonal signals to other organs to indicate the state of energy reserves and maintain the glucose, lipid, and appetite balance. But when it becomes dysfunctional (hypertrophic, inflammatory, or fibrotic, as described previously), its endocrine communication with the rest of the body becomes dangerously disrupted. In this section, we will examine the main changes in the secretion profile of diseased adipose tissue: decrease in protective adipokines, increase in harmful factors, and the consequences of these alterations on target organs.
Leptin
This is the signature hormone of adipose tissue, encoded by the LEP (or ob). Leptin is primarily secreted by white adipocytes in proportion to their lipid reserves. The more fat one has, the more leptin is produced. Its main role is to signal the state of energy reserves to the brain (particularly the hypothalamus). Elevated leptin levels indicate abundant fat stores and induce a central decrease in appetite and an increase in energy expenditure (it is a long-term satiety signal). Conversely, when fat mass decreases, leptin levels drop, which stimulates hunger and reduces energy expenditure (an energy-saving mechanism). Leptin thus links adipose tissue and eating behavior in a homeostatic loop. It also affects other organs: it regulates reproductive function (a certain leptin threshold is required for ovulation and puberty, explaining amenorrhea in women who are too thin), it modulates immunity (it is a cytokine of the IL-6 family, promoting the inflammatory response), and plays a role in regulating blood pressure (via the sympathetic nervous system).
In common obesity, we observe hyperleptinemia (excess leptin) but, paradoxically, this is associated with a preserved appetite: this is the phenomenon of leptin resistance. The brains of obese individuals no longer correctly perceive leptin’s anorexigenic signal, possibly due to alterations in the receptors or in the transport of leptin to the hypothalamus. Consequence: despite full reserves, the appetite is not properly curbed. This concept of leptin resistance is reminiscent of insulin resistance; it is a major pathological mechanism underlying obesity. In contrast, rare individuals with a mutation leading to the absence of leptin are in a state of extreme hyperphagia and morbid obesity from childhood, a condition treatable with recombinant leptin (a success story in molecular medicine). This confirms both the crucial role of leptin in weight regulation and the fact that in common obesity, the problem is not a lack of leptin but an impaired response to it.
Adiponectin
This is the other major hormone of adipose tissue, but with a very different profile. Adiponectin is produced almost exclusively by white adipocytes (and to a lesser extent by beige/brown adipocytes). It is particularly abundant in the blood (it is one of the most concentrated proteins in the circulation of a healthy individual, at around 5 to 10 µg/mL). Adiponectin increases insulin sensitivity in the liver and muscles: it promotes fatty acid oxidation, glucose uptake, and reduces hepatic glucose production. It also has anti-inflammatory and anti- -atherogenic effects (for example, it inhibits monocyte adhesion to the vascular endothelium, moderates macrophage activation, and stimulates the production of vasodilatory nitric oxide). Adiponectin is often referred to as a cardiovascular and metabolic protective factor. Notably, the more body fat increases, the more adiponectin decreases (this is the opposite of leptin).
Obese individuals have low levels of adiponectin, which contributes to insulin resistance and inflammation. Even within the general population, low levels of plasma adiponectin is predictive of type 2 diabetes and coronary heart disease. Why does adiponectin decrease as adipose tissue grows? It is believed that adipocyte hypertrophy and local inflammation inhibit its production. Visceral adipose tissue, in fact, produces much less of it than subcutaneous tissue. Thus, adiponectin is a marker of adipose tissue health: the more functional and balanced it is, the more it releases. Restoring high levels of adiponectin is an important therapeutic goal. Certain medications, such as fibrates or glitazones, increase it, as do weight loss and exercise.
Resistin
Discovered in mice as a link between obesity and insulin resistance (hence its name), resistin is a pro-inflammatory adipokine. In rodents, it is produced by adipocytes and elevated in obesity, leading to diabetes. In humans, the situation is different: resistin is primarily secreted by immune cells (macrophages) present in adipose tissue, rather than by the adipocytes themselves. Nevertheless, its blood levels are also higher in cases of obesity and may contribute to hepatic insulin resistance and systemic inflammation.
It is considered a marker of adipocyte inflammation. Its exact role in humans is less well understood than in mice, but studies link it to atherosclerosis, for example.
IL-6, TNF-α and other cytokines
These are typically immune cytokines, but adipose tissue (via its macrophages and also adipocytes for TNF) produces significant amounts of them. In obesity, up to 30% of circulating IL-6 may originate from adipose tissue. IL-6 has complex effects: acutely, it promotes lipolysis and energy expenditure (exercise); chronically, it causes inflammation and hepatic insulin resistance. TNF -α, on the other hand, was the first factor suspected in obesity-related insulin resistance: it is overproduced in adipose tissue and blocks insulin signaling in adipocytes and muscles by phosphorylating the receptor and IRS -1. Mice lacking TNF are more resistant to obesity-induced diabetes. In humans, trials of anti-TNF antibodies have not improved diabetes, perhaps because adipocyte inflammation utilizes redundant pathways. Nevertheless, adipocyte TNF-α remains an important local factor in dysfunction: it stimulates lipolysis (releasing too many free fatty acids), inhibits adiponectin, and recruits more macrophages (self-perpetuating inflammation). Other pro-inflammatory cytokines (IL-1β, MCP-1, etc. ) are also secreted by distressed adipose tissue and reinforce the inflammatory syndrome.
Other adipokines and factors
The list is long, but here are some of the most notable:
Visfatin (NAMPT): initially identified in visceral tissue (hence its name), it plays an enzymatic role in the NAD pathway and can act as a pro- -insulin (mimicking insulin on certain receptors). Its significance remains debated.
Omentin: a largely anti-inflammatory and insulin-sensitizing adipokine, produced mainly by visceral adipose tissue in lean individuals but which decreases in cases of obesity.
Apelin: a peptide produced by adipocytes (and other tissues) that improves insulin sensitivity and increases with obesity, perhaps as a compensatory mechanism.
FGF21: fibroblast growth factor 21, a metabolic hormone primarily from the liver but also from brown adipose tissue. Brown adipose tissue secretes FGF21 during thermogenesis, which acts to increase energy expenditure and insulin sensitivity overall. It is being tested as a potential drug.
Angiotensinogen: produced by adipose tissue and converted into angiotensin. It acts locally and systemically and may contribute to hypertension associated with obesity (via salt retention and vasoconstriction).
PAI-1: plasminogen activator inhibitor-1, is produced in large quantities by visceral fat, which promotes a prothrombotic state (increased risk of blood clots) in obese individuals.
Adipsin (Complement Factor D): a complement protein produced by adipocytes, involved in innate immunity. It has recently been discovered that it can improve insulin secretion by pancreatic beta cells by protecting a hormone (GLP-1) from degradation. Adipsin tends to decrease in cases of advanced obesity, which could contribute to beta cell exhaustion.
RBP4 (Retinol -binding protein 4): a retinol transport protein produced by adipocytes, elevated in obesity, and implicated in peripheral insulin resistance by disrupting insulin signaling in muscles.
Lipid signals: adipose tissue also releases specific fatty acids, ceramides, endocannabinoids, etc., which act as paracrine or endocrine signals. For example, an excess of circulating saturated fatty acids (originating from adipose tissue) can activate Toll-like receptors in the innate immune system and trigger inflammation (hence the idea that free fatty acids themselves are pro-inflammatory “lipokines”). Conversely, certain lipids produced by brown adipose tissue (oxidized fatty acids) may have beneficial effects on the liver or heart.
Exosomes and microRNAs: Adipose tissue releases vesicles (exosomes) into the circulation containing non-coding RNAs (microRNAs) and proteins that can remotely influence gene expression in other organs. For example, adipocyte microRNAs have been identified that modulate muscle muscles. This mode of intercellular communication is an emerging field.
SFRP5: This is an anti-inflammatory adipokine (Wnt pathway inhibitor) produced by healthy adipose tissue. In obesity, its expression decreases, lifting a brake on inflammation in macrophages (via Wnt5a).
This list demonstrates that adipose tissue is one of the most polyglot endocrine glands in the human body. It speaks the language of metabolic hormones, immune cytokines, growth factors, bioactive lipids, etc. Under normal conditions, this symphony of adipokines contributes to balance: leptin ensures that weight remains stable, adiponectin protects the arteries and blood sugar levels, small cytokines maintain local immune vigilance, etc. In cases of obesity, the harmony breaks down: leptin cries out but is no longer heard, adiponectin falls silent,
and the trumpets of inflammation dominate the orchestra.
In addition to these protein-based adipokines, dysfunctional adipose tissue releases excessive amounts of free fatty acids (FFAs) into the circulation, especially during the postprandial or nocturnal phases, due to its insulin resistance. These elevated FFAs have their own deleterious endocrine effects: they induce hepatic insulin resistance, decrease muscle glucose uptake (energy competition), and stimulate excessive insulin production by the pancreas (because the pancreas detects more FFA as a substrate and secretes more insulin to metabolize them—this is the concept of “glucolipotoxicity”). These FFA can also accumulate in the heart, causing cardiac disorders (cardiac lipotoxicity). Thus, diseased adipose tissue also acts by sending massive amounts of lipids into the bloodstream, imposing a burden on other organs that they should not have to bear.
Communication with target organs (brain, muscle, liver, etc.)
Adipokines exert their effects on virtually all organs. Adipose tissue is a central player in a complex communication network sometimes called the “adipoaxis,” running parallel to the classic endocrine axes. Let’s summarize the main organ-by-organ interactions:
Brain (hypothalamus)
The leptin/insulin duo informs the brain of energy reserves. When adipocytes are full, leptin (coupled with postprandial pancreatic insulin) activates anorexigenic neurons (POMC/CART) in the hypothalamus and inhibits orexigenic neurons (NPY/AgRP) . This leads to reduced food intake and increased energy expenditure (via activation of the sympathetic nervous system). The brain also receives other signals from fat cells: for example, circulating free fatty acids (released during lipolysis) can cross the blood-brain barrier and modulate the sensitivity of certain neurons to insulin or leptin.
In obesity , leptin resistance in the hypothalamus leads to a sort of “overdose” of orexigenic signals (elevated AgRP, etc.) despite caloric excess. The brain behaves as if it needs to store more. This is a major disruption in communication between adipose tissue and the brain. Conversely, in anorexia or after a severe diet, low leptin triggers very powerful hyperphagic and hypometabolic responses that complicate the stabilization of lost weight (the body “defends” its reserves). This illustrates how adipose tissue, via leptin, is an integral part of the appetite control system.
Muscles
Muscles use fatty acids as fuel, supplied in part by adipose tissue. Healthy, insulin-sensitive adipose tissue releases fatty acids at the right time (during exercise, fasting) and stores them after meals, thus preventing muscles from being overwhelmed with lipids when they don’t need them. Furthermore, adiponectin increases fatty acid oxidation in muscle and glucose uptake; it acts via its receptors (AdipoR1) to activate kinases (AMPK) that stimulate muscle metabolism. Therefore, higher levels of adiponectin mean muscles that burn fat and glucose more efficiently.
In cases of obesity (low adiponectin, high free fatty acids), muscles accumulate toxic lipids (ceramides, diacylglycerol) that disrupt insulin signaling (PKC activation, etc.) and cause muscle insulin resistance. We can thus see how dysfunctional adipose tissue induces metabolic myopathy. Conversely, a trained muscle secretes myokines (muscle IL-6, irisin, myostatin, etc.) that in turn modulate adipose tissue: exercise-induced IL-6 enhances lipolysis and may locally reduce adipose tissue inflammation, irisin promotes browning, etc.
Liver
The liver is closely linked to adipose tissue, particularly via the portal circulation (referring to the portal vein) for visceral fat. A high flow of free fatty acids from adipose tissue reaches the liver and can lead to non-alcoholic fatty liver disease (NAFLD), literally ectopic fat storage in hepatocytes, as these cells receive more fatty acids than they burn or export. Furthermore, adipokines influence liver homeostasis: adiponectin has an anti-steatotic and anti-inflammatory effect on the liver (it activates AMPK in the liver, reducing glucose and lipid production, and attenuates liver inflammation).
In obesity, the decrease in adiponectin and the influx of resistin, TNF, and IL-6, exacerbate hepatic insulin resistance: the liver becomes less sensitive to insulin, continues to produce too much glucose and VLDL, while storing more fat. The result is the well-known metabolic fatty liver that often accompanies visceral obesity. The adipose tissue–liver axis is therefore central to metabolic syndrome. Some refer to adiponectin as “liver insulin” because its action there is so beneficial, and to resistin/TNF as “liver anti-insulins.” On the other hand, the liver influences adipose tissue via the production of factors (e.g., IGF-1, liver-derived sex hormones, etc.), but this is less direct.
Adipose Tissue Itself (Autocrine/Paracrine)
It should not be forgotten that most of these adipokines also act on the adipose tissue itself. For example, leptin has autocrine effects on adipocytes (it can limit lipid synthesis), adiponectin may act locally to regulate preadipocyte differentiation, and local TNF-α directly inhibits triglyceride accumulation in adipocytes (it promotes lipolysis). Thus, the secretion profile of adipose tissue modulates its own plasticity: too much TNF and not enough adiponectin, and preadipocytes no longer differentiate properly (inhibiting hyperplasia); too much leptin locally could limit fat storage. This local system is complex and still under study.
Pancreas (β cells)
Adipose tissue sends signals to the pancreas. For example, leptin can act on the pancreatic islets by slightly inhibiting insulin (a potential anti-obesity effect, as less insulin equates to less fat storage). In obesity, elevated leptin maintains a certain pressure to limit hyperinsulinism, but central leptin resistance can counteract this. Adiponectin, through its effects on insulin sensitivity, reduces the burden on β-cells and may have a direct cytoprotective effect on these cells.
Conversely, excess fatty acids from adipose tissue cause “lipotoxicity” in β-cells, reducing their function and leading to apoptosis. It is also suspected that an adipokine such as adipsin (mentioned above) helps β-cells by increasing the duration of action of GLP-1, which would boost glucose-dependent insulin secretion. In advanced obesity, the combination of insulin resistance (forcing β-cells to overwork) and lipotoxicity (weakening β-cells) often leads to pancreatic exhaustion, and thus to type 2 diabetes. Adipose tissue plays a dual role: it is because of it that the body needs a lot of insulin (peripheral resistance), and it is also because of it that β-cells fail (due to lipid and cytokine damage). Thus, dysfunctional adipose tissue, through the hormonal feedback loop, drives the pancreas to failure.
Kidneys
Elevated leptin levels in obesity stimulate the renal sympathetic nervous system and may contribute to increased sodium reabsorption, thereby leading to hypertension. Furthermore, visceral and perirenal adipose tissue produces angiotensin II locally, enhancing vasoconstriction. Hyperinsulinemia (secondary to insulin-resistant adipose tissue) also stimulates salt reabsorption by the kidneys. All of this makes adipose tissue a contributing factor in obesity-related hypertension. Furthermore, obesity is is accompanied by expanding perinephric and renal perivascular fat, which may promote local inflammation and renal fibrosis via adipokines. This gives rise to the concept of renal lipotoxicity.
Heart and blood vessels
Adiponectin has protective effects on the myocardium (anti-apoptotic, and prevents pressure-induced cardiac hypertrophy). A decrease in adiponectin in obese individuals may thus contribute to diabetic cardiomyopathy. Excess leptin has pro-hypertrophic effects on the heart and may promote arrhythmias via the sympathetic nervous system. At the vascular level, an unhealthy adipokine profile (high TNF, IL-6, angiotensin, low adiponectin) causes endothelial dysfunction, inflammation of the vascular wall, and the progression of atherosclerosis. Visceral adipose tissue is notably implicated in the development of coronary atherosclerosis (via elevated IL-6, which increases CRP, and angiotensin II, which promotes hypertension). Thus, cardiovascular diseases linked to obesity are largely mediated by adipokines. It can be said that atherosclerosis is a distant consequence of adipose tissue inflammation.
Immune System
Adipose tissue, via leptin and adipokines, influences overall immune function. Leptin, for example, stimulates the innate immune system (activation of macrophages and neutrophils) and directs adaptive immunity toward a pro-inflammatory Th1 response rather than a Th2 response. Very thin or malnourished individuals (low leptin) often have weakened immunity, whereas obese individuals have an overactive immune system, albeit in an aberrant manner (chronic inflammation but reduced response to specific infections). This has been observed with COVID-19: obesity was a risk factor for severe disease, possibly due to a low-grade inflammatory state amplifying the cytokine storm. Adiponectin, on the other hand, moderates certain macrophage functions and can increase anti-inflammatory cells. Adipose tissue also secretes components of the complement system (adipsin, etc.), contributing to humoral immunity. We are beginning to understand that adipose tissue could be an immune organ in its own right, modulating the balance between tolerance and inflammation in the body.
We could continue with other organs (bones , skin, endocrine glands, etc.)… For example, leptin influences the gonadotropic axis (too-low levels inhibit fertility, while very high levels could also disrupt spermatogenesis or ovarian function), and subcutaneous fat, as mentioned, helps the skin defend itself, etc. But let’s focus on the essentials: adipose tissue communicates with nearly all of the body’s body. It sends signals regarding energy status, inflammation, growth, and vascularization, and in return receives hormonal signals (insulin, sex hormones, cortisol, adrenaline…) that modulate its activity. This multi-system integration makes adipose tissue a key organ for maintaining internal balance (homeostasis).
Thus, dysfunctional adipose tissue can be described as a pathogenic endocrine organ: instead of fulfilling its regulatory role, it sends aberrant signals to the body that induce or exacerbate metabolic diseases. From a therapeutic perspective, one strategy aims to restore a healthier adipokine profile (for example, researchers have sought ways to increase adiponectin, or leptin analogs to overcome resistance, etc., with limited success so far). However, the sustainable normalization of the adipocyte endocrine profile often requires reducing fat mass and restoring plasticity (through weight loss or interventions targeting tissue quality; see the following section).
Summary
Given this cascade of consequences, one could say that obesity is first and foremost a disease of adipose tissue. It is the dysfunction of this tissue (rather than excess weight per se) that triggers the associated pathologies. Moreover, some people can be obese without having many apparent metabolic problems—a condition known as metabolically healthy obesity— these are often people whose adipose tissue, though abundant, has remained relatively functional (less visceral fat, adipocytes not too large, little fibrosis, good hyperplasia, adipokines not too dysregulated). Conversely, people of moderate build may have highly dysfunctional adipose tissue (e.g., a lot of visceral fat, ectopic infiltration) and develop diabetes or a heart attack. This is why we must emphasize the concept of adipose tissue quality.
Today, some researchers suggest that risk assessment in an overweight patient should not be limited to weight or BMI, but should look for markers of adipocyte function: fat distribution (waist circumference, imaging), levels of key adipokines (adiponectin, leptin), indicators of excessive lipolysis (elevated free fatty acids), etc. For example, a low adiponectin-to-leptin ratio in the blood is associated with insulin resistance and may reflect impaired adipose tissue.
In summary, dysfunctional adipose tissue is the central theme in the pathogenesis of obesity. It converts chronic energy surplus into a cascade of pathogenic signals that affect nearly all organs. The good news is that if we can “resynchronize” this tissue—primarily through fat loss, but also through other interventions—many of these abnormalities regress. This is the basic principle of obesity treatment obesity: improving adipose function by reducing the load and modifying lifestyle to make the remaining fat healthier. In the next section, we will examine the clinical implications of all this and the current or future therapeutic strategies for managing dysfunctional adipose tissue.
Clinical and Therapeutic Implications
Knowledge of adipose tissue biology has concrete implications for the management of obesity, diabetes, and metabolic diseases. Understanding that this is a disease of the adipose tissue guides therapeutic strategy: it is not enough to reduce fat mass; we must also attempt to improve its function. In this section, we will discuss some practical implications: how to assess a patient’s adipose tissue (beyond weight), how lifestyle can influence adipose tissue health, which treatments directly or indirectly target adipose tissue, and what the limitations and prospects of these approaches are.
Clinical Assessment of Adipose Tissue: Beyond Weight
In clinical practice (or even in sports coaching), BMI is only a starting point. Two people with the same BMI may have very different adipocyte profiles. It is therefore important to assess fat distribution and certain biological markers to refine the picture.
In short, no single test is sufficient, but a set of indicators (e.g., a patient who is not very overweight but has a large waist circumference, low adiponectin, high triglycerides, low HDL, etc.) will indicate damage to the visceral adipose tissue.
From the patient’s or athlete’s perspective, the assessment should highlight that not all weight gain is the same. It is better to gain weight in the form of muscle or peripheral subcutaneous fat than in the form of abdominal fat. Similarly, when it comes to weight loss, reducing waist circumference is a priority.
In sports medicine, we may encounter athletes with a normal BMI but high levels of visceral fat (particularly in certain strength sports where cardio is not a major focus). These individuals may benefit from tests such as DEXA or MRI to alert them to their actual body composition.
In medicine, a detailed understanding of adipocyte distribution has led to the concept of phenotypes: “metabolically healthy” vs. “metabolically unhealthy.” We could further refine this by identifying an “adipocyte fibrosis phenotype” or “inflammatory phenotype” via fat biopsies. In practice, routine fat biopsies are not performed, except for research or when a rare pathology is suspected, but perhaps one day?
Lifestyle, exercise, and improving adipocyte health
As is often the case, lifestyle interventions are the cornerstone of treatment, and they have profound effects on adipose tissue.
For patients, it is important to emphasize that even without significant weight loss, exercise improves body composition and metabolic health. We often see patients lose few pounds but many inches from their waistline thanks to exercise: this means that visceral fat has melted away and lean body mass has increased, which may not show up on the scale but makes a huge difference to health. Any increase in physical activity (walking, sports, strength training) is beneficial for “treating” problematic adipose tissue.
In short, lifestyle can radically transform radically alter the behavior of adipose tissue. A balanced diet and regular exercise transform “diseased” adipose tissue into “healthier” tissue, smaller, more insulin-sensitive, and less inflammatory. The positive impact on health stems largely from this. One could almost say that the goal of dietary and physical management is to restore adipocyte plasticity: to restore adipose tissue’s ability to absorb energy without causing inflammation, and its ability to release it when needed.
Therapeutic Approaches Targeting Adipose Tissue
In addition to lifestyle measures, there are several medical approaches (pharmacological or surgical) that aim either to reduce fat mass or to modify the functioning of adipose tissue. Here are the main ones:
Weight-loss medications: The arsenal of anti-obesity drugs has long been limited, but recent advances have transformed the landscape.
GLP-1 analogs (liraglutide, semaglutide, etc.), administered via injection, were originally antidiabetic drugs that have proven highly effective in inducing significant weight loss (–10% to –15% on average) . They work by sharply reducing appetite and delaying gastric emptying. Indirectly, by reducing caloric intake, they create a deficit in adipose tissue, thereby triggering its depletion. It has been observed that, beyond fat loss, these molecules improve insulin sensitivity and reduce systemic inflammation, suggesting a beneficial effect on adipocyte function (perhaps via a decrease in leptin and a relative increase in adiponectin). For example, one year of semaglutide can virtually “cure” type 2 diabetes in an obese patient, meaning that their adipose tissue has significantly reduced in size and regained a proper hormonal balance. These medications are revolutionizing the management of severe obesity, as they offer significant weight loss without surgery, which permanently reduces the burden on adipose tissue.
SGLT2 inhibitors (gliflozins) are also antidiabetic drugs that induce modest weight loss (–2% to –3%) by causing glucose to be excreted in the urine. They do not specifically target adipose tissue, but by inducing a caloric deficit, they slightly stimulate lipolysis. They may slightly reduce visceral fat. Not a cure-all for obesity, but useful in diabetic patients for whom a little weight loss helps.
β3 receptor agonists (such as mirabegron, a urological drug) have been tested for their ability to activate brown fat. A high dose of mirabegron can increase resting energy expenditure by approximately 100 kcal/day via brown adipose tissue, which is not insignificant. However, these high doses can also cause tachycardia and hypertension (mirabegron at standard doses has less of this effect). It is not (yet) approved for obesity, but it is a promising avenue. Other substances such as ephedrine (an adrenergic stimulant) have been used in the past to increase thermogenesis, but at the cost of negative cardiovascular effects. Pharmaceutical research is attempting to find molecules that activate brown fat without stimulating the heart, or that induce selective browning.
Thiazolidinediones (TZDs), such as pioglitazone, are worth considering. Used to treat type 2 diabetes, they act by activating the PPAR-γ transcription factor, which is the “master regulator” of adipogenesis. Consequently, these drugs increase adipocyte hyperplasia and lipid storage in subcutaneous adipose tissue. Clinically, patients on TZDs gain a little weight (3–5 kg on average), but paradoxically, their insulin sensitivity improves significantly. This is explained by water retention and by the fact that fat is redirected to adipose tissue (safe storage) and leaves the liver and muscles. This treatment can be viewed as “nourishing adipose tissue” by increasing its healthy storage capacity. Imaging studies have confirmed that pioglitazone increases subcutaneous fat mass but reduces hepatic fat and improves adipose tissue composition (smaller adipocytes, higher adiponectin levels). Unfortunately, TZDs have side effects (water retention, risk of heart failure, etc.) that limit their use, and they are not specifically approved for obesity without diabetes. Nevertheless, they have demonstrated the principle that obesity can be treated by improving the quality of adipose tissue rather than by eliminating it. This is an interesting paradigm shift: instead of seeking to destroy fat, we transform it into active “ active ‘good fat’ and redistribute it more effectively. This doesn’t result in weight loss on the scale, but the patient’s metabolic health improves significantly. In the future, we might see more targeted molecules that leverage this concept without the drawbacks of TZDs.
Anti-inflammatories: Given the role of adipocyte inflammation, anti-inflammatories have been tested to improve insulin sensitivity. For example, salsalate (an older salicylate ) or JNK inhibitors, but clinical results are modest and they are not commonly used. Cytokine antagonists (anti-TNF, IL-1) have not shown sufficient improvement in diabetes to justify their use (except perhaps IL-1 in certain types of diabetes, but this is a special case). It is likely that once the process is established, blocking one cytokine is not enough, as others take over. So no “adipose tissue anti-inflammatory pill” for now. However, high-dose omega-3 fatty acids or certain flavonoids (such as quercetin) are being studied for their anti-inflammatory effects on adipose tissue: for example, a dasatinib+quercetin combination has been tested to eliminate senescent (inflammatory) cells from the tissue. But this is still experimental.
Agents Modulating Fibrosis: There is no specific medication to reduce adipose fibrosis. Certain molecules such as pirfenidone (an antifibrotic used in pulmonary fibrosis) could theoretically be effective, but they have significant systemic effects and are not indicated for obesity. Metformin has some indirect anti-fibrotic properties via AMPK and could limit adipose tissue fibrosis by improving oxygenation (it promotes angiogenesis in certain models). But this is not its primary effect.
In summary, recent pharmacological research has focused on overall weight reduction (GLP-1) and metabolic redistribution (TZD). The former target the brain axis (appetite regulation), while the latter target adipose tissue directly. The future may lie in combining both approaches, which is already happening: new “combined” molecules such as tirzepatide (a dual GLP-1/GIP agonist) induce even greater weight loss (–20% in trials), which will definitively relieve adipose tissue of its excess. One can imagine that subsequently, a more refined PPAR-γ modulator would be used to consolidate the functionality of the remaining adipose tissue.
Bariatric surgery: This is currently the most effective treatment for morbid obesity. By drastically restricting food intake (and/or absorption), surgery (sleeve gastrectomy, gastric bypass, etc.) induces significant weight loss (often >25% of initial weight). This results in massive adipose tissue loss over 12 to 18 months. Surgery also has hormonal effects (increased endogenous GLP-1, etc.) that enhance weight loss and maintain satiety. From the perspective of adipose tissue:
After surgery, a dramatic reduction in the size of adipocytes is observed, and sometimes even in the number of adipocytes (there may be a certain degree of adipocyte apoptosis since the loss is rapid, and potentially autophagy). A regression of hepatic steatosis (within a few months) and an almost immediate improvement in insulin sensitivity (often even before significant weight loss, thanks to hormonal changes). A decrease in systemic inflammation (CRP and IL-6 levels drop), and an increase in adiponectin. In some patients, the hyperandrogenism associated with polycystic ovary syndrome improves, fertility returns, a sign that the gonadal axis is responding to the reduction in harmful adipose tissue.
In the longer term, adipose tissue rebuilds itself differently: often the proportion of visceral fat is much lower afterward. Fat distribution can return to near-normal levels. However, in cases of weight regain, it has been observed that visceral fat often returns quickly (possibly be because the fibrous structure of the initial visceral adipose tissue remains and facilitates refilling).
Surgery, by removing adipose tissue, demonstrates the extent to which all metabolic abnormalities can improve to the point of disappearing in a formerly obese individual. Of course, this is only offered for severe or complicated obesity, as the procedure is highly invasive. But the message is: the more we can sustainably reduce adipose tissue, the better it is for overall health.
Experimental therapies: Here are a few futuristic approaches:
Brown adipose tissue transplantation: in mice, implanting additional brown fat improves metabolism. In humans, we’re not there yet, but researchers are studying how to differentiate stem cells into brown adipocytes and transplant them.
Genetic engineering: targeting key genes in adipose tissue. For example, one approach would be to block the collagen-binding protein (ATX enzyme) to limit fibrosis, or to modulate HIF-1. However, there is concern about potential adverse effects. For example, overexpression of HIF-1 promotes obesity…
Senescent cells: With age, adipocytes become senescent and secrete large amounts of cytokines (SASP). Eliminating these cells using senolytics (molecules that preferentially kill senescent cells) is being explored to improve adipose tissue function in elderly obese individuals.
Microbiota: The gut microbiota influences lipid storage. Fecal transplants from lean donors to obese individuals have shown slight improvements in insulin sensitivity insulin. The microbiota communicates with adipose tissue via metabolites (short-chain fatty acids, etc.). Perhaps manipulating the microbiota (prebiotics, probiotics) might also modulate adipocyte function. For example, certain probiotics reduce adipocyte inflammation in some models.
Local devices or treatments: Cryolipolysis locally destroys adipocytes using intense cold (for aesthetic purposes), but it is unclear whether removing subcutaneous fat in this way affects metabolic health . Surgical liposuction also removes fat, but studies have shown that removing 5 kg of subcutaneous fat does not improve metabolic parameters at all because visceral fat mass is not affected, and adipocyte physiology remains unchanged. Sometimes, even after liposuction, the body tends to regain what was removed, sometimes in deeper deposits. This underscores the need to address the cause (caloric excess) and adipocyte function, not just remove superficial fat.
Conclusion
Adipose tissue, once viewed as mere inert and unsightly padding, is now recognized as a central organ of physiology whose diversity, plasticity, and dysregulation profoundly influence human health. We have seen that there are several types of fat (white, brown, beige) with complementary functions, and that the anatomical distribution of fat deposits is a key factor, often making the difference between well-tolerated excess weight and harmful excess.
The plasticity of adipose tissue allows the body to adapt to energy fluctuations: by storing more or fewer lipids, recruiting new adipocytes, altering cell size, or even converting white adipocytes into brown adipocytes, or by remodeling its vascularization and immune environment. This plasticity ensures healthy expansion. When it reaches its limits (extreme hypertrophy, unresolved inflammation, rigid fibrosis), adipose tissue enters a state of dysfunction and becomes the starting point for a cascade of metabolic disorders (insulin resistance, dyslipidemia, hepatic steatosis, etc.).
Obesity can be understood, from a mechanistic perspective, as the story of adipose tissue that is first overburdened, then overwhelmed, and finally diseased. In a domino effect, diseased adipose tissue sends erroneous signals to the body—whether it be a brain misled about the state of reserves, a liver clogged with fat, inflamed arteries, or an exhausted pancreas. Thus, the concept ofadipopathy takes on its full meaning: it is not so much the quantity of fat that is the problem as the loss of its functionality.
In practice, this knowledge translates into monitoring waist circumference, a profound change in eating habits and behaviors, encouragement of physical activity, and an awareness that even modest weight loss can yield disproportionate benefits if it targets the right fat compartment. We also gain a better understanding of certain conditions such as metabolically healthy obesity (where adipose tissue is still functioning) or, conversely, diabetes in non-obese individuals (where adipose tissue is too limited or dysfunctional). This calls for a more personalized approach to overweight : treating “the person and their adipose tissue,” rather than a BMI number.
In conclusion, adipose tissue is a dynamic endocrine and immune organ, endowed with an astonishing capacity for remodeling, at the heart of a delicate balance between health and disease. If it remains flexible and well-regulated, it protects the body from energy imbalances. But if it becomes rigid and inflamed, it becomes a source of systemic diseases. The good news is that, even when it has been damaged, adipose tissue retains the capacity to improve: by reducing excess calories, mobilizing its reserves through exercise, and thanks to emerging therapies, its functions can be partially restored. In other words, the story of adipose tissue is not set in stone. This notion carries a message of hope for patients: by addressing the causes (lifestyle, environmental factors) and using the appropriate medical tools, it is possible toreverse the course of metabolic syndrome by giving adipose tissue the means to return to healthier functioning.
The challenge for the coming years will be to continue unraveling the intricate mechanisms of this adipocyte plasticity, in order to develop even more targeted and effective interventions, such as selectively stimulating brown/beige adipose tissue, increasing adipose tissue capacity without promoting obesity, or identifying reliable circulating biomarkers of adipose tissue health. In any case, recognizing adipose tissue as a key organ must be the foundation of change: we are not fighting (only) against fat for aesthetic reasons; we are caring for an organ on which our metabolic balance largely depends.
References
- Arner, E., Westermark, P. O., Spalding, K. L., Britton, T., Rydén, M., Frisén, J., Bernard, S., & Arner, P. (2010). Adipocyte Turnover: Relevance to Human Adipose Tissue Morphology. Diabetes, 59(1), 105–109.
- Cypess, A. M., Lehman, S., Williams, G., Tal, I., Rodman, D., Goldfine, A. B., Kuo, F. C., Palmer, E. L., Tseng, Y.-H., Doria, A., Kolodny, G. M., & Kahn, C. R. (2009). Identification and Importance of Brown Adipose Tissue in Adult Humans. New England Journal of Medicine, 360(15), 1509–1517.
- De Fano, M., Bartolini, D., Tortoioli, C., Vermigli, C., Malara, M., Galli, F., & Murdolo, G. (2022). Adipose Tissue Plasticity in Response to Pathophysiological Cues: A Connecting Link between Obesity and Its Associated Comorbidities. International Journal of Molecular Sciences, 23(10), 5511.
- Gilani, A., Stoll, L., Homan, E. A., & Lo, J. C. (2024). Adipose Signals Regulating Distal Organ Health and Disease. Diabetes, 73(2), 169–177.
- Liu, F., He, J., Wang, H., Zhu, D., & Bi, Y. (2020). Adipose Morphology: A Critical Factor in Regulation of Human Metabolic Diseases and Adipose Tissue Dysfunction. Obesity Surgery, 30(12), 5086–5100.
- Lopez-Yus, M., Hörndler, C., Borlan, S., Bernal-Monterde, V., & Arbones-Mainar, J. M. (2024). Unraveling Adipose Tissue Dysfunction: Molecular Mechanisms, Novel Biomarkers, and Therapeutic Targets for Liver Fat Deposition. Cells, 13(5), 380.
- Mo, Y.-Y., Han, Y.-X., Xu, S.-N., Jiang, H.-L., Wu, H.-X., Cai, J.-M., Li, L., Bu, Y.-H., Xiao, F., Liang, H.-D., Wen, Y., Liu, Y.-Z., Yin, Y.-L., & Zhou, H.-D. (2024). Adipose Tissue Plasticity: A Comprehensive Definition and Multidimensional Insight. Biomolecules, 14(10), 1223.
- Peirce, V., Pellegrinelli, V., & Vidal-Puig, A. (2016). Adipose Structure (White, Brown, Beige). In R. S. Ahima (Ed.), Metabolic Syndrome (pp. 369–396). Springer International Publishing.
- Peng, Y., Zhao, L., Li, M., Liu, Y., Shi, Y., & Zhang, J. (2024). Plasticity of Adipose Tissues: Interconversion among White, Brown, and Beige Fat and Its Role in Energy Homeostasis. Biomolecules, 14(4), 483.
- Rutkowski, J. M., Stern, J. H., & Scherer, P. E. (2015). The cell biology of fat expansion. Journal of Cell Biology, 208(5), 501–512.
- Saito, M., Okamatsu-Ogura, Y., Matsushita, M., Watanabe, K., Yoneshiro, T., Nio-Kobayashi, J., Iwanaga, T., Miyagawa, M., Kameya, T., Nakada, K., Kawai, Y., & Tsujisaki, M. (2009). High Incidence of Metabolically Active Brown Adipose Tissue in Healthy Adult Humans. Diabetes, 58(7), 1526–1531.
- Sakers, A., De Siqueira, M. K., Seale, P., & Villanueva, C. J. (2022). Adipose-tissue plasticity in health and disease. Cell, 185(3), 419–446.
- Stenkula, K. G., & Erlanson-Albertsson, C. (2018). Adipose cell size: Importance in health and disease. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 315(2), R284–R295.
- Virtanen, K. A., Lidell, M. E., Orava, J., Heglind, M., Westergren, R., Niemi, T., Taittonen, M., Laine, J., Savisto, N.-J., Enerbäck, S., & Nuutila, P. (2009). Functional Brown Adipose Tissue in Healthy Adults. New England Journal of Medicine, 360(15), 1518–1525.
- Weisberg, S. P., McCann, D., Desai, M., Rosenbaum, M., Leibel, R. L., & Ferrante, A. W. (2003). Obesity is associated with macrophage accumulation in adipose tissue. Journal of Clinical Investigation, 112(12), 1796–1808.
- Zwick, R. K., Guerrero-Juarez, C. F., Horsley, V., & Plikus, M. V. (2018). Anatomical, Physiological, and Functional Diversity of Adipose Tissue. Cell Metabolism, 27(1), 68–83.